Fl1 RNA-syntes och processning
advertisement

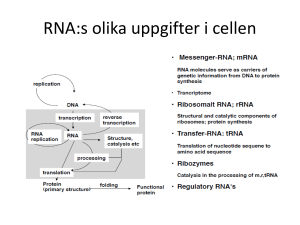
L1: RNA Synthesis and Processing Outline L1: RNA Synthesis and Processing • Transcription in Prokaryotes • Eukaryotic RNA Polymerases and General Transcription Factors • Regulation of Transcription in Eukaryotes • RNA Processing and Turnover Vad är en gen? What was a gene yesterday? gen promotorer signalpeptid transkriptionsstart RBS transkriptionstermineringssignal TAA TAG TGA ATG GTG TTG strukturell gen transkriptionsenhet What is a gene today? “A DNA segment that contributes to phenotype/function. In the absence of demonstrated function a gene may be characterized by sequencee, transcription or homology” – Wain et al, Genomics, 2002 “A locatable region of genomic sequence, which is associated with regulatory regions, transcribed regions and/or other functional sequence regions” – Pearson, Nature, 2006 “Alternatively spliced transcripts all belong to the same gene, even if the proteins that are produced are different” – the Gene Sweepstake team, 2003 The encode project-home work! A gene is a genomic sequence (DNA or RNA) directly encoding functional product molecules, either RNA or protein In the case that there are several functional products sharing overlapping regions, one takes the union of all overlapping genomic sequences coding for them This union must be coherent – i.e., done separately for final protein and RNA products – but does not require that all products necessarily share a common subsequence or just The gene is a union of genomic sequences encoding a coherent set of potentially overlapping functional products Nature 2007 Jun 14;447(7146):799-816 1 E. coli RNA polymerase Transkription i prokaryoter 1. Initiering: RNA-polymeras binder promotor, strukturella förändringar hos promotorpolymeraskomplexet (”bubbla”), initial transkription 2. Elongering: konformationsförändringar (håller hårdare om templatet), uppvindning av DNAt, RNA-syntes, ”proof-reading” 3. Terminering: Rho-oberoende terminatorer (inneboende terminatorer = hårnålsstrukturer), Rho-beroende terminatorer DNA footprinting (Part 1) Sequences of E. coli promoters 16-19 bp RNA polymeras: α2ββ´ω+ σ (ca 465 kDa) - sigmafaktorn påverkar DNA-bindande egenskaperna hos polymeraset - bindningsstyrkan till olika promotorer kan skilja 106 x - transkription initieras utan primer 5-9 bp • Startsignaler för RNA-syntes • Stark promotor: hög affinitet för RNA-polymeras • Konstitutionell promotor: alltid påslagen • Reglerbar promotor: går att slå på och stänga av DNA footprinting (Part 2) Sigmafaktorer (σ) i E. coli Gene Factor Use -35 sep -10 rpoD σ70 general TTGACA 16-19 bp TATAAT rpoH σ32 heat shock CCCTTGAA 13-15 bp CCCGATNT rpoN σ54 N-metabolism CTGGNA 6 bp TTGCA fliA σ28 (σF ) motility/chemotaxis CTAAA 15 bp GCCGATAA rpoE σE heat shock rpoS σS stress response 2 Transcription by E. coli RNA polymerase (Part 1) Transcription by E. coli RNA polymerase (Part 2) • • Structure of bacterial RNA polymerase (after ca 10 bp) ~ 12-14 bp Transcription termination in Prokaryotes RNA synthesis continues until the polymerase encounters a termination signal. The most common signal is a symmetrical inverted repeat of a GC-rich sequence followed by seven A residues. Alternatively, transcription of some genes is terminated by a specific termination protein (Rho), which binds extended segments of single-stranded RNA. Transcription termination Transcription termination (E. coli) Promoter RNA polymerase DNA RNA ”Hairpin” termination signal 5´ 5´ Other mechanism: Termination via Rho protein (hexamer) Pause-site Rho 3 Transkriptionsreglering Genreglering efter transkriptionsinitiering • Prematur transkriptionsterminering s.k. attenuering; ex. trp • Skiljer på positiv och negativ reglering • Positiv reglering: transkriptionsfaktor (aktivator) måste binda in till promotorn för att transkription skall ske - hjälper RNA-polymeras att binda till DNAt (rekrytering) - allosterisk verkan av steg efter polymerasinbindning • Antiterminering = en typ av positiv transkriptionsreglering vid vilken transkripionsfaktorer (proteiner) binder till RNApolymeras och modifierar det så att det kan läsa igenom speciella termineringssites - används av fager och i vissa operon • Negativ reglering:repressorprotein binder till operator och hindrar transkription - hindrar RNA-polymeras att binda till promotorn - alternativt håller kvar RNA-polymeraset Metabolism of lactose Negative control of the lac operon Positive control of the lac operon by glucose Reglering av laktos-operonet (E. coli) Catabolite Activator Protein (CAP) Promotor -35 Cykliskt Promotor= AMP ”Landningsplats” för (cAMP) RNA-polymeras CAP -10 Lac I S.D. 5´ Lac-repressor (Lac I) Laktos eller IPTG (syntetisk analog) DNA -10 Lac Z Lac I mRNA Lac I mRNA-start RNA-pol. -35 3´ cAMP/CAPbindningsställe Operatorsekvens S.D. 5´ Lac Z β-galaktosidas: Spjälkar laktos till galaktos och glukos Lac Y S.D. Lac Y Laktos-permeas: Reglerar införsel av laktos Lac A S.D. mRNA Lac A 3´ Thio-galaktosid-acetylas: Bryter ned ej klyvbara laktos-analoger Frånvaro av laktos: • Lac-repressorn binder till operator-sekvensen • Transkriptionen blockeras; inget β-galaktosidas-enzym (eller något av de andra enzymerna) produceras Närvaro av laktos (eller IPTG): • Lac-repressorn kan inte binda till operator-sekvensen • Transkriptionsker; β-galaktosidas-enzym (och alla de andra enzymerna) produceras Närvaro av laktos (eller IPTG) samt låga halter av glukos: • Halten av cAMP stiger • cAMP-CAP-komplex bildas som kan binda uppströms om promotorn • cAMP-CAP-komplex främjar transkriptionen (vägleder RNA-polymeraset) • Mer β-galaktosidas-enzym produceras 4 Eukaryotic RNA Polymerases and General Transcription Factors Structure of yeast RNA polymerase II Eukaryotic cells have three nuclear RNA polymerases that transcribe different classes of genes. They are complex enzymes, consisting of 12 to 17 different subunits each. They all have 9 conserved subunits, 5 of which are related to subunits of bacterial RNA polymerase. Eukaryotic RNA Polymerases and General Transcription Factors Formation of a polymerase II preinitiation complex in vitro (Part 1) RNA polymerase II is responsible for synthesis of mRNA and it has been the focus of most transcription studies. Unlike prokaryotic RNA polymerase, it requires initiation factors that (in contrast to bacterial σ factors) are not associated with the polymerase. General transcription factors are proteins involved in transcription from all polymerase II promoters. About 10% of the genes in the human genome encode transcription factors, emphasizing the importance of these proteins. Promoters contain several different regulatory sequence elements. Promoters of different genes contain different combinations of promoter elements, which appear to function together to bind general transcription factors. Formation of a polymerase II preinitiation complex in vitro (Part 2) Model of the polymerase II preinitiation complex 5 RNA polymerase II/Mediator complexes and transcription initiation The ribosomal RNA gene is transcribed by RNA polymerase I Initiation of rDNA transcription Transcription of RNA polymerase III genes Identification of eukaryotic regulatory sequences A eukaryotic promoter 6 The SV40 enhancer Action of enhancers DNA looping The immunoglobulin enhancer Electrophoretic-mobility shift assay Chromatin immunoprecipitation (Part 1) 7 Chromatin immunoprecipitation (Part 2) Structure of transcriptional activators Examples of DNA-binding domains Action of transcriptional activators Action of eukaryotic repressors Regulation of transcriptional elongation (Part 1) 8 Regulation of transcriptional elongation (Part 2) Regulation of transcriptional elongation (Part 3) Regulation of Transcription in Eukaryotes Regulation of Transcription in Eukaryotes The packaging of eukaryotic DNA in chromatin has important consequences for transcription, so chromatin structure is a critical aspect of gene expression. Chromatin can be modified by: Modifications of chromatin structure play key roles in the control of transcription in eukaryotic cells. • Modifications of histones • Interactions with HMG (high mobility group) proteins • Rearrangements of nucleosomes Actively transcribed genes are in relatively decondensed chromatin, which can be seen in polytene chromosomes of Drosophila. Histone acetylation (Part 1) Patterns of histone modification • Histone modification: • The amino-terminal tail domains of core histones are rich in lysine and can be modified by acetylation. Transcriptional activators and repressors are associated with histone acetyltransferases (HAT) and deacetylases (HDAC), respectively. • 9 Chromatin remodeling factors Chromatin remodeling factors are protein complexes that alter contacts between DNA and histones. They can reposition nucleosomes, change the conformation of nucleosomes, or eject nucleosomes from the DNA. Regulation of Transcription in Eukaryotes To facilitate elongation, elongation factors become associated with the phosphorylated C-terminal domain of RNA polymerase II. They include histone modifying enzymes and chromatin remodeling factors that transiently displace nucleosomes during transcription. Regulation of Transcription in Eukaryotes: DNA methylation • • DNA methylation is another general mechanism that controls transcription in eukaryotes. Methyl groups are added at the 5-carbon position of cytosines (C) that precede guanines (G) (CpG dinucleotides). RNA Processing and Turnover Most newly-synthesized RNAs must be modified, except bacterial RNAs which are used immediately for protein synthesis while still being transcribed. rRNAs and tRNAs must be processed in both prokaryotic and eukaryotic cells. Regulation of processing steps provides another level of control of gene expression. Processing of ribosomal RNAs Processing of transfer RNAs (Part 1) 10 Processing of transfer RNAs (Part 2) Processing of eukaryotic messenger RNAs (Part 1) Processing of eukaryotic messenger RNAs (Part 2) Formation of the 3´ ends of eukaryotic mRNAs Splicing of pre-mRNA RNA Processing and Turnover: Splicing Three sequence elements of pre-mRNAs are important: Sequences at the 5′ splice site, at the 3′ splice site, and within the intron at the branch point. Pre-mRNAs contain similar consensus sequences at each of these positions. Splicing takes place in large complexes, called spliceosomes, which have five types of small nuclear RNAs (snRNAs)—U1, U2, U4, U5, and U6. They are complexed with six to ten protein molecules to form small nuclear ribonucleoprotein particles (snRNPs). 11 Alternative splicing in Drosophila sex determination Alternative splicing of Dscam 12x48x33x2=38016 combinations RNA Processing and Turnover: RNA editing Editing of apolipoprotein B mRNA RNA editing is processing (other than splicing) that alters the protein-coding sequences of some mRNAs. It involves single base modification reactions such as deamination of cytosine to uridine and adenosine to inosine. Editing of the mRNA for apolipoprotein B, which transports lipids in the blood, results in two different proteins: Apo-B100 is synthesized in the liver by translation of the unedited mRNA. Apo-B48 is synthesized in the intestine from edited mRNA in which a C has been changed to a U by deamination. RNA Processing and Turnover: RNA degradation Aberrant mRNAs can also be degraded. Nonsense-mediated mRNA decay eliminates mRNAs that lack complete open-reading frames. When ribosomes encounter premature termination codons, translation stops and the defective mRNA is degraded. Ultimately, RNAs are degraded in the cytoplasm. Intracellular levels of any RNA are determined by a balance between synthesis and degradation. Rate of degradation can thus control gene expression. RNA Processing and Turnover rRNAs and tRNAs are very stable, in both prokaryotes and eukaryotes. Bacterial mRNAs are rapidly degraded, most have half-lives of 2 to 3 minutes. In eukaryotic cells, mRNA half-lives vary; less than 30 minutes to 20 hours in mammalian cells. Short-lived mRNAs code for regulatory proteins, levels of which can vary rapidly in response to environmental stimuli. mRNAs encoding structural proteins or central metabolic enzymes have long half-lives. 12 RNA Processing and Turnover mRNA degradation Degradation of eukaryote mRNAs is initiated by shortening of the poly-A tails. Rapidly degraded mRNAs often contain specific AUrich sequences near the 3′ ends which are binding sites for proteins that can either stabilize them or target them for degradation. These RNA-binding proteins are regulated by extracellular signals, such as growth factors and hormones. Degradation of some mRNAs is regulated by both siRNAs and miRNA. 13