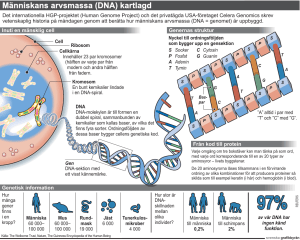
Faglambda Kortfattad labbeskrivning
advertisement

.
,
.
Nedanföljer en mycketkortfattadbeskrivningav
Konstruktionoch screeningav genbibliotek
OBS! i labhandledningen
"LaboratoryDNA Science"användsplasmidenpBLU mm vi
använderistället plasmidenpUC 18.
e
1. E.ro" lM 101 odlasÖ~
2. KL YVNIN G
BakteriofagA'sDNA ochplasmidenpUC 18klyvs med restriktionsenzymen
Hind m
och BarnHl i separatarör enligt följande;
15 J.Il BakteriofagÄ DNA+ 3J.1l1OxY+tangobuffert+ 1.5 J.IlBam 81+ 1.5 J.Il Hindm + 9
J.Il
vatten
8 J.lIplasmid pUC 18+ 3J1l1OxY+tangobuffert+ 1.5J.1l 'am Hl+ 1.5J1l Hindm+ 16J1l
vatten
Rörenintuberasi 3'PC vattenbadi 30-60minuter
somhinnerkontrolleraratt klyvningenfungeratgenomatt analyseraen del av
. De
klyv produkternapå en agarosgel somfärgasin medEtBr.
. Alla funderaröverhur resultatetbordebli på gelen.
(8
~:8
3. LIGERING
4 J.11
kluven Ä.DNA+ 4J.Llkluven pUC18 + 2 J.Llligasbuffert + 9 J.LlVatten + 1 J.Llligas
4. TRANSFORMATION
E.coli 1M 101 görs kompetenta(SO\mMCaCh behövs)
Förbered LB plattor med ampicillin till en slutkonc pi ca 100 Jlglml, IPTO till en
slutkonc pi ca 1 mMlmI och X-gal till en slutkonc pi ca 30 Jlglml per platta (tex SOJ1lav
en 2OmglmlX-gal lösning till en platta med 30 mi LB).
.
1ynga:
Bricka (el dyl):
Papperslager
(filterpapper):
.
LJ
----
Nytt nylonmembran:
m
U rspnmgsny1onmembran:
.;",?
Filterpapper
(Whatman
3mm):
.
-+
-+
Kortaregenomgångav laborationeni lab-salen
Lösnlo2sberednbul:
De Dature rings b uffert
Neutraliseringsbuffert
Prehybridiseringsbuffen
Hybridiseringsbuffert
6. DENATURERA. PREHYBRIDISERA(ÖD). HYBRIDISEBMON) och
FRAMKALLA.
Buffert!
Buffert 2
Buffert 3
r.;;:
TV Ä]T A MEMB RANET
FRAMKALLA MED ANTI-D I G OXI GENIN ANTIKR O PP K O N JU G ERA T TILL
ALKALISKTFOSFATAS
+ SUBSTRAT (ni tro blue tetrazollum salt och ]tpbospbate)
7.SÅTT
N KULTURERAV
8. PLASMIDPREPARATIONPÅ DE PO
RESTRlKTIONSENZYMKL
YVNING
~
A KLONERN
~
TABLE
TABLE~1.
~1.
BlololY
810101Y
CommonRestrlctlon
Enzymes
UsedInMolecullr
MIc:roorpnls18
Enzyme Rec:olI'ltlonSlte
Acinetobocter calcoacetlcus
AccI
b:Z
fcnignDN4
~~~
5' G
G TLiTLAC
T~
Ae
S'
eA~TG
CA~T6
BamHI 5'
5'GIG
GIGA
AT~
Te~
Bocillusamylollque{oclms
H
l
Cut DNA with r8IIridian
=-~
811&ym8le.g..
HiIdIt
3"
((TAG
(CTA
Escherlchlacoli RY13
EcoRI
5' GIAAT~
eTTA
5' AGCn
A G CTT
5'
TT(G
nCG
5' G
GT
T ~)
~)I C) AC
Ae
5'
CTTA
5'GIAAT~C
HoemophilusInfluenzaeRd
Hlndlll
H. in/fuenzaeserotypeC.1160
Hlncll
m
CAC)eTG
(AmerG
5' C
e!! ~
GIACGT
IACG'T(e
G
5' e C
e CIGGG
ClGG6
5'C
GGGlccc
G
G GIe ( (
provldenc1a
stuartn164
Pst!
Serratlo
marcescens
Smal
Stophylococcus
auteus]A
Sau3A1 51G
5'IGAT~
T~
eTA
CTA
Xmal
tIC (G
cG G
5' ((
- - - - G_1GGGCC
Xonthomonasmalvoceol1lm
The DNA to be cloned can be obtalned
5'
G
I
!I.
'
r' 9.
-
JO
.~ ~:r
'.
::J:r
,5'
"-a,""'"
--
by puriftcatioD
of chromosomal DNA from ceDa.vtruses.or other plamlds or by selective ampl1ficatioD of DNA sequences by a
technlque knoWD as polymerase chain react1on (PCR).
(PCR Is explained further in Chapter 17.) 80th the vector
and the foreign DNA are eleaved with restrlction enzymes
(Figure 5-11). Restrlct10D enzymes recognIze a spedftc
paIlndromlc
sequence and make a staggered rot. wh1ch
generatessticky enda.or a blUDtcut. whlch generates
.
~:
blUDt ends (see Table 5-1).
~~.:
. sequencethat can be eleavedby many restriction
Most doning
vecton
have
::".
~,
enzymes. called the multiple
clowns
slte. Llgation or
the rector with the DNA fragments generates a molecule
~:~
capableof repllcattngthe Insertedsequence
called
tecomblnant
DNA (see FIgure 5-9). The total number of
r~mblnant
vecton obtained when elonlng all the frag..~ts that result from eleavage of chromO8Omal DNA Is
t<'
, ~'.'
',:(r. J61ownas agenomIe Ubrary becausethere shouldbe
- ~ .:~ }c:ast one representative
,
,
.
of esch gene In the library.
<i:~M
..'E.
" ' ~te;natJve approach to eloning the gene for a protein
cotlvert the roRNA for the protein Into DNA using a
'\
11
~
,,
-
"
>~~
\.
,
enzymecalled reversetranscrlptase(RNA-
'
;.,~ " ndent DNA polymerase) to produce a complementary
.:~P:~JcDNA).
t;
~~sed
"
A cDNA Ubrary
representa the genes that
asmRNAIn a parttcularceD.
'pi~recomblnant
DNA Is then transformed Into a bao~~~.ost.
usualJy B. cou. and the plasmld-contalnlng
.
..
. e selected for antJbloUc reslstance (e,g., ampi-
,
,
'~.'
. .'
. ~~
'.r:
.
tante).Thelibrarycan thenbescreened
to Ond
. oJ, elone po5Sesslng the desIred DNA
.
"
,
fragment.
.~eenlng technlquescan beosedto Identlfythe
~~o~tainJng the approprtate recomblnant DNA.
The multiple elonlng site used for Insertlng the foreign
DNA is often part of the lacZgeneof the lIJCoperan. 1n8eI'tion of the foreign DNA into the lacZgeneinaettvatesthe
gene (aeting almost like a transposon)and prevents the
plasmld-d1rected synthesis of ~ctos1dase
in the ~
lent cell. whteh results in whIte bactertalcolontesInstead
of blue colontestf p-galactosidasewere able to eleavean
---'-.- ~U1UWUVU~
-L._-~-"---
aVI11UVIIDUW
Genet1cenglneer1ng has been osed to !solate and
expressthe genes for useful proteins In bacterta. yeast,
Dr' even Insect cells such as 1nsul1n.Interferon. growtb
hormones. and Interleu1dn. Large amountl of pure
immunogen for a vacclne can be prepared without the
needto wort with the Intact d1sease
organisms.
45
The developmentof a vacclne agalnst hepatit1sB virus
representsthe first successof recomblnant DNA vacc1nes
approvedfor human use by the ns. Pood and Drug
Adm1n1stration.The hepatitls B surface antigen is ~
ducedby the yeastSaccharomyces
cerevfslae.
In the future
lt may be sufficient to Inlett plasmid DNA capable of
expressIngthe deslredimmunogen (DNA vacclne)Into an
tndtvldualto let the hast cellsexpressthe immunogenand
generatethe immune response.RecomblnantDNA technology has also becomeessentialto laboratory dtagnosls.
forenslescience.agrlculture. and many other d1sctpllnes.
dIagnoses.
Alberts B et al: Molecularblologyof theceU,ed 3. New York. 1994.
Garlaod.
Cooper GM: TheceD:a molecu1ar
approoch.Washington, 1997.
American Societyfor MlcroblolOl}'.
Lehninger AL. Nelson DL. FaxMM: Prfnclplesof blochemlstry.ed
2. New York. 1993. Wortb.
LewinB: GenesVI. Oxford. England. 1997. Oxford University.
Lodlsh H et al: Molecularcell blology.ed 4. New York. 2000. WH
Freeman.
Stryer L: Bfochemlstry.ed 4. New York. 1995. Preeman..
VoetD, VoetJG: Bfochentfstry.ed 2, New York. .199S. Wl1ey.
Watson ]D et al: MoleculJ1rbfology oj the gene,ed 4, Menlo Park.
Callf. 1987, Benjamln-Cummlnp.
Weigel LM et al: Genetic analyslsof a high-\evel vancomycinresistant isolateof Staphylococcusaureus, Scfmce302: l 5691571.2003.
.
'.;:";';.":;"i.~';'
T',.
l
,
. ",
,.,
"
",(
',H.
8
.
I.
C
mRNA
flGURE 5-1. A. The lactose operon Is transcrlbed IS I poIy-
mRNA 1
~
I
.....
t
~~
T
. ,
induc8r
8
.
:.
+.
CI#
D
,
J
ndu.,
meraseat the operatorslte (O).C.Therepressor,
comptexed
with
formation change in the repressor. The lat operon 15 thus transcrlbed It I low tevel. D, Eschltrlchla coU is IfOwn In I poot'
.
.
cAN#
ATP
1
I
I
.
~
~ ~
.-.
:.1._---.
. ~~.
y
T
..T1i:)
cAHI
to the promoter rellon and bindini of the IctIYe repressor to the
operator sequence, betause no Inducer Is aval!able. The result
wlU be that the loc operon will not be transcnbed.ATP,
adenosine
trlphosphate; CAp, catlballte
adenoslne rnonophosph8te.
llne-aetlvltor
protein; cAMP, cycUc
AtP
'
e
medium In the presente of !actose IS the urban source. 80th the
Inducer and the CAP-cAMPcomplu: are bound to the promoter,
which Is fully 'turned on: and I high tevel of 1ocmRNAIs t,.,..
scrlbedandtranslated.
E,Growthof E.coli in I poor medium
without tlctOseresuttsIn the binding of the CAP-cAMPcompla
...
CAP
and
the Inducer, does not recognlze the operator becauseof I can-
I
Rep.-
M,
~
mANA
I
CP) and trans-
00, permelse
Inducer, becluse the repressor competea with the RNA poly-
Y
'9
IIiIII
t
~galactosldase
~
-; p
.
RNA (m RNA) from the promoter
proteins:
Icetylase
W.The
IDC
Ilene
encodes
therepressorprotein. ..The
Ilctose
operon
15nottrlnscrlbed
Intheabsente of In IltolactoM
Repqnor
mANA
messenger
Into three
~
Ilransgaloclo6ida11on
ladDI8
..
T
,
c:Istronk
-.
-
"tlted
J
Repr-..or
I
Ad8nyIoI.
R.-
E
8
New DNA Is syntheslzedsemiconservadveJy, using
both strandsof the parental DNA astempiates.New DNA
to correct any errors that were made. Durlng log-phue
growth in rlcb medium, many Loltlatlonsof chromO8Omal
syotheslsoccursat arowlng rorks and proceedsbidlrepUcattonmay occur before cell division.ThIs process
rudonalJy. ODe strand (the leading strand) Is copled produces a series of nested bubbles of new daugbter
contlnuousJyIn the 5' 10 3' direction. whereasthe other
cbromosomea,eacb with its pair of growth forks of new
strand (the lagging strand) must be synthesizedas many
DNA synthesls. The polymerase moves down the DNA
piecesof DNA uslng RNA primers (Okazaki'sfragments). strand, Incorporating the appropriate (complementary)
The legglng.strandDNA must be extendedin the 5' 103'
nucleotide at each position. ReplJcationIIIcompletewhen
direction as Its template becomes available. Then the
the two repUcatlon forks meet 180 degrees from the
pieces are Ugated together by the eI1%)TIDe
DNA ligase origtn. The processof DNA replication puts great torsional
(Figure 5-2). To malntain the high degree of accuracy
strain on the chromosomal circle of DNA; this strain il
required for repUcation. the DNA poJymerasesposse88 reUevedby topolsomerasa (e.g.. gyrase).which super"proofreading" functions. whlch allow the enzyme 10
coil the DNA. Topolsomerases
are essentiaI10the bacterla
confirm that the appropriate nueleottdewas insertedand
and are targets for the qulnolone antibiotk:a.
.
)
j
~
:'
c
BACTERIAL
GENETICS
~
Al' ~
0'
'\J
FlGURE 5-2. Batterlal DNA replkatlon. New DNA synthesis occurs at
growing forks and proceeds bidirectlonally. DNA synthesis progresses
in the 5' to 3' directlon contlnuosly (leading strand) or In pieces (lagglng
strand). Assuming It takes 40 minutes to complete one round of repll.
catlon and assumIng new Initiation every 20 minutes, initiation of DNA
synthesis precedes cell division. Multiple growlng forks may be Initiated
in a cell before complete septum formation and cell division. The
daughter cells are -born pregnant."
T~_~~!!~tlonalControi
REGULATIONOF GENE EXPRESSlON
8
Bactertahavedevelopedmechanlsmsto adaptqulckly and
cfficiently to changes in concentratlons oc nutrlents in
thelr envlronment.The batteris turn on a completeset oc
enzymeswhen necessaryand avoldmaking the enzymeor
enzymesoc a pathway when the substrateIs absent.
Flrst. the organization oc the genesof a blochemlcal
puthwuy Into an operon, with appropriate genetic
controi mechanisms. sHows coordlnated productlon of
the nt..'Cessary
enzymes to response to a nutrltlonal stImu-
lus. Seeond.the transcriptlon of the gene Is regulated
directly by repressorproteins (which bind to operators)
In responseto nutrltlonal signals within the cell. ThIrd.
the rate of protein synthesisby the rtbosomecan regulate
transcrtptlon 10 prokaryotes. The absente of a nuclear
membraneIn prokaryotesallows the ribosometo bind to
the mRNA as It Is belog traoscrlbed from the DNA.
TRANSCRIPTlONAL
REGULATION
Initiation of transcription may be under positiveor negativecontroI. Genesunder negative controi are expressed
r.
;4-.
~
-~
'
;
i",':'\.';;1'::",~<':'f~/j;~'~
c~~ .'c. .. '
~ '~ ,>: l:~ ~1
c
. .c,~ . .rI":;~
'
.
' "
c
c
...
"
"
" cc
"'
c c:c
c
'
-'
CHAPTERJ
MultipleGrowingFn
8
...c-'c
c
>",-
c
.
c
.
c
;'.~
c
c
c
.
c
-~ .i~l'.' '", ~;0r'"
':'::"""::",,~';/':":
.c\'~
. ~~
\;':'t~
.~
.
c
c
c
"'" .c""c;c ~~
':r"~~~
"'."
unlessthey are switchedoff by a repressor protein. This
repressorprotein preventsgeneexpresslonby binding to a
specificDNA sequencecaned.the operator, maldng lt
Impossiblefor the RNA polymeraseto Initiate transcrtptlon at the promoter.Inversely.geneswhose expressionis
under posidve contral are not transcr1bedunless an
actlveregulator protein. canedan apoinducer, is present.
The apolnducer binds to a speclfic DNA sequenceand
assiststhe RNA polymerasein the In1tlatlon stepsby an
unknown mechanism.
Operanscan be inducible or repressible. Introductlon of a substrate (inducer) into the growth medium
may induce an operon to Increasethe expressionof the
enzymesnecessaryfor Its metabolism.An abundante of
the end products (co-repressors) of a pathway may
signal that a pathway should be shut down or repressed
by reducing the synthestsof its enzymes.
Thelactose(lac)operonresponsiblefor the degradation
of the sugar lactoseis an Induc1bleoperonunder positive
and negative regulation (~ Figure 5-1). Normally the
bacteria use glucose and not tactose.In the absente of
lactose the operan is repressedby the bInding of the
repressorprotein to the operator sequence.thus lmped1ng
the RNA polymerasefunction. In the absenteof glucose.
however,the addition of lactosereversesthis repression.
Full expresstonof the lac operan aIsarequ1resa proteInmediated.positive-controlmechanlsm.In B. coU.a protein
calledthe catabol1tegene-activatorprotein (CAP)forms a
complex with cycl1cadenosinemonophosphate(cAMP).
acquiring the abil1tyto bind to a speclflcDNA sequence
present In the promoter. The CAP-cAMP complex
enhancesblnding of the RNA polymeraseto the promoter.
thus allowing an increasein the frequency of transcrlption lnitiation. The CAP-cAMPcomplexmay Incressethe
operon transcription by protein-protein interaction with
the RNA potymeraseor by protein-DNAiIlteraction.
The tryptophan operan (trp operon) contains the
structural genes necessaryfor tryptophan blosynthes1s
and is under dual transcrtptional contral mechanlsms
(FIgure5-3). Although tryptophan is essentialfor protein
synthesis.too much tryptophan in the cell can be toxic:
thereforeits synthestsmust beregulated.At the DNA level
the repressorprotein is activatedby an increasedintracellular concentratIon of tryptophan to prevent transcriptIan. At the protein synthestsleveI.rapid translation of a
"test peptide" at the beglnning of the roRNA in the presence of tryptophan promotes the formation of a doublestrandedloop in the RNA. which terminatestranscription.
The sameloop is formed if no protein synthesisis occurring. a situation In which tryptophan synthesis would
slmllarly not be requlred. This regulates tryptophan
synthesis at the roRNA level in a process termed atteDuadon. In which roRNA synthes1sis prematurely
terminatecl.
37